Please note that this newly restructured FAQ page includes new and revised questions related to the 8th edition of the Guide for the Care and Use of Laboratory Animals (NRC 2011). If you have a question not addressed in our FAQs, please email accredit@aaalac.org. For further guidance, please refer to AAALAC International's Position Statements or Guidance Statements.
I have read that accreditation assessments are based on three primary standards, the 8th Edition of the Guide for the Care and Use of Laboratory Animals (Guide), NRC 2011; Guide for the Care and Use of Agricultural Animals in Research and Teaching (Ag Guide), American Dairy Science Association®, the American Society of Animal Science, and the Poultry Science Association 2020; and the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes, Council of Europe (ETS 123). Our animal care and use program is located in the United States. Does this mean that we now have to follow ETS 123 for our program to be accredited?
The Council on Accreditation has used all three documents for several years and their adoption by AAALAC International's Board of Directors as primary standards signifies the importance of these performance-based guidelines in the accreditation process. AAALAC’s Rules of Accreditation state "The accreditable unit shall observe any and all statutes and governmental regulations which bear upon animal care and use including, but not limited to, the prevailing standards of sanitation, health, labor and safety of the jurisdiction(s) in which it is located." The scope of applicability of ETS 123 is limited to the member countries of the Council of Europe that have voluntarily ratified the Convention, and thus it is not a prevailing standard in the United States.
There may be value, however, in reviewing AAALAC's collation of performance standards pertaining to cage/pen space excerpted from the Guide, Ag Guide and Appendix A of ETS 123 as each guidance document provides sound recommendations regarding housing environments for a variety of species. For example, while the Guide is silent on appropriate housing for ferrets, ETS 123 provides excellent guidance which would be useful for institutions around the globe. ETS 123 may also be used for assessments of programs located outside of Europe that have established an institutional policy to follow this standard in addition to local regulations and guidelines. AAALAC encourages institutions to make animal care and use determinations based on regulatory and funding requirements, overlaid with a performance approach that enhances animal welfare and quality science.
If animals owned by our institution are housed at another institution for production of antibodies, breeding transgenic colonies or quarantine, are these activities considered part of our AAALAC accredited program? Will the activities, facilities and program at the contract site be evaluated during our site visit?
AAALAC follows animal ownership in terms of defining who is responsible for animals at an offsite program. In the examples described, your institution has confirmed ownership of the animals; therefore, if the contractor's program is not AAALAC accredited, a description of the animal care and use activities and facilities provided by the contract facility must be included in your AAALAC Program Description. The contract facility would also be visited during your triennial site visit and contractual agreements must provide for the review of the contracted facility by AAALAC site visitors. Specifically, you would need to include a detailed description of the relevant portions of the contractor's program (e.g., facilities, husbandry, veterinary care, IACUC or comparable oversight body review, etc.) for those elements of their program that pertain to or impact the care and use of your animals. The site visit team would also evaluate the portions of the contractor's facility(ies) that are relevant to your animals. If the contract facility is a significant distance from your main facility, this may have bearing on the site visit logistics, and you should contact the AAALAC International office to discuss the situation. During the scheduling of the dates of your site visit, the Council member leading the visit must be made aware that contract services are provided off-site by a non-accredited program. If you initiate a contract with a program that is not accredited between accreditation site visits, this change should be reflected in your Annual Report to AAALAC.
If the contractor's program is separately accredited by AAALAC, it will not be necessary to visit that facility during the site visit to your institution. Also, if your institution does not own the animals, but just owns the data (or intellectual property) that result from studies or procedures conducted using those animals, AAALAC will not visit the non-accredited site. The Spring 2003 issue of the Connection Newsletter contains an in-depth article on this particular subject (click here to download) and is available for downloading from the AAALAC International Web site. As the scope of animal ownership and responsibility may vary depending on a country's regulations, policy and/or agency guidance, AAALAC International strongly encourages institutions be familiar with local prevailing standards as well as AAALAC's approach.
We use a variety of invertebrates in our research program and I heard somewhere that they have to be included as part of our accredited program. Can you clarify this for me?
The following statement is from the Rules of Accreditation as found on the AAALAC website:
"All animals used or to be used in research, teaching or testing at accreditable units are to be included and evaluated in accordance with the standards set forth in Section 2 of these Rules. This includes traditional laboratory animals, farm animals, wildlife, and aquatic animals. Nontraditional animals, inclusive of invertebrate species, are also included where they are relevant to the unit's mission."
Of particular concern to Council, vis-a-vis invertebrate colonies, is the inclusion of their review when such inclusion would benefit the program as a whole, or their exclusion would potentially jeopardize the overall program mission. Also, Council acknowledges the need to evaluate management of invertebrate colonies when those colonies are within a core animal facility and therefore subject to creating, or being subject to, potential adverse influences from activities in adjacent spaces. Possible areas of evaluation could include:
While AAALAC does require that invertebrates used as a part of the research mission of the institution be included as part of the animal care and use program, there are many ways in which that can be accomplished. Many institutions do not have a formal protocol for invertebrate work, but notify the Institutional Animal Care and Use or Ethics Committee or comparable oversight body (IACUC/OB) via letter about the work they will be doing with these species. Or the oversight may be a Standard Operating Procedure (SOP), policy statement, guideline, etc., that is reviewed by the IACUC/OB and delineates the committee's responsibility over the use of invertebrates and provides guidance to investigators using invertebrates. Some of the issues the document may want to include are listed above. The intensity of oversight by the IACUC/OB may depend on such things as the species of invertebrate being used and the type of procedures being performed. Site visit teams will generally not review research activities involving lower level invertebrates such as zooplankton, sea slugs, nematodes, or mosquitoes, but will review research with higher level invertebrates such as cuttlefish, squid or octopi. For example, if procedures are being conducted on a higher level invertebrate, an IACUC/OB approved protocol may be an appropriate requirement.
The guidance from AAALAC is, of necessity, flexible because a site visit team would be very interested in the IACUC/OB oversight of octopus research, as compared to oversight of work with zooplankton or nematodes. For more information on the care and use of cephalopods, please see the AAALAC International Reference Resource (click here).
We are considering using a former employee as our nonaffiliated member. They are no longer affiliated with our program and they are familiar with the type of research we perform. Are there any concerns with this approach?
The Guide for the Care and Use of Laboratory Animals (Guide), NRC 2011, does offer some guidance on the issue of what should be some of the traits of the nonaffiliated (public) member. The Guide states this person should represent the general community interests in the proper care and use of animals, should not be laboratory-animal users, and not be affiliated with the institution or be members of the immediate family of a person who is affiliated with the institution. The OLAW/ARENA Institutional Animal Care and Use Committee Guidebook (which is an AAALAC International reference resource) expands on this by stating that the nonaffiliated member can bring significant value to the committee by bringing a non-institutional perspective to the research endeavor. For programs with an NIH Assurance, OLAW provides some additional guidance in their frequently asked questions section of their website (http://grants.nih.gov/grants/olaw/faqs.htm#IACUC_1) by stating that "the unaffiliated member should have no discernable ties or ongoing affiliation with the institution, and may not be a member of the immediate family of a person who is affiliated with the institution. Immediate family includes parent, spouse, child and sibling. Appointment of an individual who is unambiguously nonaffiliated is the best way to fulfill the letter and spirit of this provision." Using a former employee as the nonaffiliated member is certainly a gray area that might come into question during a site visit based on the above guidance, especially in context of the comments about the value of that member bringing a non-institutional perspective to the research endeavor and not having any discernable ties or ongoing affiliation with the institution. While not specifically prohibited, the Institutional Official should give special consideration to the use of a former employee as a nonaffiliated member to ensure that both the intent and the spirit of the Guide are being met.
My institution is located outside the United States and there is no government requirement for our institution to develop a mechanism for reporting animal welfare concerns, though we have good lines of communication within our institution so that we are certain that any concerns would be discussed with management. The Guide for the Care and Use of Laboratory Animals (NRC 2011) describes a number of specific elements of a reporting system. Are all of these required?
AAALAC International recognizes that the regulatory requirement for a defined method for reporting animal welfare concerns is primarily limited to the United States. However, as noted in the Guide, it is the responsibility of everyone associated with the animal care and use program to ensure animal welfare. In some instances, this may involve having to make a formal report regarding a welfare concern. Providing a method by which such reports can be made anonymously and without fear of reprisal, and enhancing staff awareness of the importance and means of reporting animal welfare concerns through training, the posting of signage, and other communication modalities are critical elements of the reporting program. It is AAALAC's expectation that such reports will be investigated by the appropriate oversight body (e.g. , the IACUC or comparable oversight body) and that any necessary corrective actions will be taken. In addition, AAALAC International should be informed of the results of the investigation and any subsequent corrective measures.
An investigator at my institution regularly performs survival surgical procedures on rodents in her laboratory. Our IACUC has reviewed and approved these activities. The recently updated Guide states that the laboratory should be dedicated only to surgical activities during the time that surgery is performed. Does this mean that all other activities in the laboratory must stop while surgery is being performed? What is AAALAC’s opinion?
The Guide for the Care and Use of Laboratory Animals (NRC 2011) states, “For most survival surgery performed on rodents and other small species such as aquatics and birds, an animal procedure laboratory, dedicated to surgery and related activities when used for this purpose and managed to minimize contamination from other activities conducted within the room at other times, is recommended." AAALAC International acknowledges that limiting nonsurgical activities in the laboratory may help to minimize contamination of the surgical area. However, AAALAC recognizes that minimizing contamination during surgery may be achieved by considering several factors. The specific location of the surgical area within the laboratory should promote the proper conduct of sterile technique, and to the extent possible, it should be isolated from other activities in the laboratory. The surgical area should be dedicated for that purpose while surgery is performed. Other factors that may impact the risk of contamination include the invasiveness and complexity of the surgical procedure, duration of surgery, and the nature of other non-surgical activities conducted in the laboratory (i.e. their likelihood of increasing the risks of surgical contamination). For complex or long procedures, or if the layout of the laboratory does not permit a suitable dedicated surgical space, it may be advisable to temporarily stop other laboratory activities, thereby dedicating the laboratory to surgery in order to maximize the potential for a good surgical outcome. For minor surgeries of short duration, conducted in a suitable area within the laboratory, it may be acceptable to allow other laboratory activities to continue if they do not jeopardize aseptic technique. The investigator, IACUC (or comparable oversight body) and veterinarian should evaluate surgical areas to ensure they are appropriate.
Our IACUC receives protocols that require use of drugs or other chemicals not formulated for clinical use, which we understand to be characterized as non-pharmaceutical-grade compounds. We would appreciate AAALAC's guidance on the distinction between pharmaceutical- and non-pharmaceutical- grade compounds and how to evaluate this aspect of these protocols.
A pharmaceutical-grade compound is defined as any active or inactive drug, biologic or reagent, for which a chemical purity standard has been established by a recognized national or regional pharmacopeia (e.g., the U.S. Pharmacopeia (USP), British Pharmacopeia (BP), National Formulary (NF), European Pharmacopoeia (EP), Japanese Pharmacopeia (JP), etc.). These standards are used by manufacturers to help ensure the products are of the appropriate chemical purity and quality, in the appropriate solution or compound, to ensure stability, safety, and efficacy.
AAALAC International acknowledges that in an animal care and use program non-pharmaceutical-grade compounds often are necessary for scientific research. Where the use of non-pharmaceutical-grade substances may be essential for the conduct of science, the goal of the IACUC (or comparable oversight body (OB)) should be to consider the health and well-being of the animals while aiding the researcher in minimizing potentially confounding experimental variables and maximizing reproducibility of the research. The Council on Accreditation will apply a performance approach to its assessment of the use of non-pharmaceutical-grade compounds, and will expect that the IACUC/OB has established acceptable criteria for use of such compounds within the institution and for review and approval of their use.
AAALAC distinguishes between two scenarios when considering the use of non-pharmaceutical-grade compounds:
Clinical Use - compounds used for the clinical treatment of animals and to prevent or reduce/eliminate animal pain or distress. Whenever possible, pharmaceutical-grade compounds must be used.
Research Use - compounds used to accomplish the scientific aims of the study. If available, and suitable, pharmaceutical-grade compounds are preferred.
In all cases when non-pharmaceutical-grade preparations are used, AAALAC International expects investigators and the IACUC/OB to consider the following factors:
Our AAALAC accredited Veterinary College is struggling over whether or not there is a requirement for client-owned animals to be covered under a protocol since they are used for teaching purposes for veterinary students during their clinical rotations, or when they become part of a research study.
AAALAC International has had a long standing policy of following animal ownership as a mechanism for determining inclusion in the accredited animal care and use program. This was highlighted in the Spring 2003 Connection Newsletter that described the AAALAC International Rules of Accreditation regarding contract facilities and how ownership determines responsibility (click here to download). The concept of ownership also applies to Colleges of Veterinary Medicine where client-owned animals are brought in for care. Even though there are some teaching and research activities involved with this scenario, the animals are not owned by the institution and so therefore AAALAC does not consider them as part of the accredited program. For animals that are owned by the College of Veterinary Medicine being used for research, teaching or other purposes (e.g., blood donors), we would expect the Institutional Animal Care and Use Committee (IACUC or comparable oversight body) to provide oversight and for the animals to be covered by a standard operating procedure or protocol that is reviewed and approved by the IACUC/OB. There is nothing that prevents an institution from establishing some sort of IACUC/OB oversight mechanism for client-owned animals; on the contrary, it would reflect a high level of institutional commitment, but AAALAC does not require it. For institutions that also hold a U.S. Public Health Service Assurance, see additional information regarding this topic at http://grants.nih.gov/grants/olaw/faqs.htm#App_8.
Recognizing the critical impact the transportation of research animals has on the quality of research and that the welfare of the animals being transported is governed by a number of regulatory agencies, what are AAALAC International's expectations to ensure humane transport of research animals?
AAALAC recognizes that the continuation of humane transportation of animals is vital to the research enterprise. However, this can be a stressful experience for the animals. The total experience of the animal during transport is influenced by many factors. It is expected that the parties involved are aware of and comply with all applicable regulatory requirements for the transportation of animals and the critical importance of ensuring that the animals are treated humanely at all times during the journey. Please refer to AAALAC International's Reference Resources (http://www.aaalac.org/resources/reference-resources/) for additional guidance.
The following points should be considered:
Security: The consignor of the transportation containers and arrangements should ensure that the possibility of damage, misdirection and breach of biosecurity is reduced to a minimum by the use of suitably recognized reputable transport operators and sub-operators. Procedures should be in place to prevent theft or adulteration of transportation containers.
Species specific transportation: Containers and transport vehicles should provide appropriate environmental conditions for the species being transported and personnel should have appropriate knowledge relative to animal biosecurity to maintain the animals' health status.
Transportation Logistics: Special attention should be given to the mode of transport and the journey times should be minimized as much as possible. Journey plans should be in place with alternate plans available in case of disruption of the original plan. The receiving institution should be informed about the itinerary and any changes in the timetable or any deviations from the plan which occur during transport. The receiving institution should have arrangements in place to receive the animals and trained personnel are available at the receiving institution to ensure the animals are removed from the shipping containers and inspected in a timely manner.
Commercial carriers: When commercial carriers are used, they should be appropriately licensed, and they should use well-designed animal transport vehicles which are capable of maintaining appropriate environmental conditions for the species being transported, which are documented through appropriate record keeping. The drivers must be trained in the transport of animals and the regulations thereof. When non-commercial carriers are used they and their vehicles should be approved by the IACUC /OB.
Planning for unexpected events: In case of an unexpected event, everyone in the transport chain should have a common understanding of contingency plans and knowledge of emergency contact persons who are available to respond during each segment of the journey. A clear emergency plan should be available and all the relevant information about contact persons, containment of the animals, and other relevant information should follow the animals on the crate or containers.
Intra-institutional transport: When animals are being transported within an institution, the IACUC/OB should approve the vehicle(s) and the personnel who perform this function, taking into account the relevant points described in this FAQ. Responsibility of the IACUC/OB: The IACUC/OB (or comparable oversight body) and veterinarian should evaluate transportation of animals to ensure compliance with regulatory expectations as well as attention to the animals' well-being.
In looking at the 8th edition of the Guide, there is a significant amount of new information regarding the enrichment program. It appears that the enrichment program at our institution now needs to be in writing and should be reviewed by the IACUC, researchers and veterinarian on a regular basis. In addition, the Guide recommends that personnel responsible for animal care and husbandry should receive training in the behavioral biology of the species under their care. Will AAALAC require all these different parties to participate in this review and should we implement a training program for staff regarding laboratory animal behavior?
Environmental enrichment is an important method of improving the well-being of many laboratory animal species and may be accomplished by the provision of stimuli, structures and resources that facilitate the expression of species-appropriate behaviors. Environmental enrichment, as one component of an IACUC (or comparable oversight body)-approved behavioral management program, should be provided in a consistent manner across the animal program, with due attention to personnel and animal safety. Personnel should be made aware of the enrichment program as one aspect of the overall training program; if it appears that the enrichment program has not been implemented properly, this may indicate inadequate training. Similar to their role in providing front-line observation for clinical illnesses, personnel responsible for daily care should be adequately familiar with normal animal behavior such that abnormal behavior may be recognized and reported. Implementation of environmental enrichment should also take into account the scientific goals of the study for which the animals are used; enrichment should be considered an independent variable and, thus, suitably controlled. While the Guide implies that a written environmental enrichment program should be in place, AAALAC site visitors will focus their attention on the IACUC's review of the enrichment program, documentation of the review, and implementation of the program. AAALAC expects that the enrichment program will be reviewed regularly by the IACUC, and that the IACUC adequately represents the research community and veterinarian(s) at the institution in the review of enrichment program.
The 2011 Guide for the Care and Use of Laboratory Animals states that "Single housing of social species should be the exception...." We house our animals in a variety of configurations and would like further explanation as to AAALAC's perspective and expectations regarding social housing.
The Guide states that "Social animals should be housed in stable pairs or groups of compatible individuals unless they must be housed alone for experimental reasons or because of social incompatibility." It is understood that research objectives, the health condition of the individual animal, and/or the challenges associated with the social behavior of some species, strains and/or sex of the animals may preclude social housing. Per the Guide "Not all members of a social species are necessarily socially compatible. Social housing of incompatible animals can induce chronic stress, injury, and even death. In some species, social incompatibility may be sex biased; for example, male mice are generally more prone to aggression than female mice, and female hamsters are generally more aggressive than male hamsters." For some species, such as rabbits of both sexes, developing and maintaining social hierarchy may entail antagonistic interactions between pair or group members resulting in superficial wounds or more serious injuries. The research and welfare benefits and consequences of both affiliative and aggressive species-typical natural social behavior must be considered. In cases where single housing is justified, the Guide states that "it should be limited to the minimum period necessary, and where possible, visual, auditory, olfactory, and tactile contact with compatible conspecifics should be provided. In the absence of other animals, enrichment should be offered such as positive interaction with animal care staff and additional enrichment items or addition of a companion animal in the room or housing area." In general, however, pair or group housing is recommended for social species (see AAALAC International's Position Statement on Social Housing, https://www.aaalac.org/accreditation-standards/position-statements/#social).
AAALAC recognizes that there is a spectrum of social experience that can be made available to an animal based on the species, health, and use of the animal. These experiences are important because, when properly managed, they can significantly enhance the welfare of the animal. Full time social housing is the optimum manner to provide social experience. However, when full time housing with conspecifics is not possible, whether due to social incompatibility, veterinary concerns or scientific necessity, other social experiences should be considered such as part time access (e.g., overnight, when the animals are between studies, defined periods of time during the day, etc.) to full contact with conspecifics or protected contact that allows interaction through a mesh panel, grooming bars or other type of perforated barrier on either a part or full time basis. In this manner, the social experience of the animal occurs as a normal aspect of the animal's housing environment or as a separate activity that occurs outside of the primary enclosure, such as in a play yard, exercise cage, animal holding room aisle or facility corridor, etc. The staff responsible for the day-to-day management and oversight of the social experience of the research animals should be well versed in recognizing aggressive and affiliative behaviors of the various species in their care to provide for rapid identification and any necessary intervention.
The Institutional Animal Care and Use Committee (or comparable oversight body) and veterinarian should periodically review the strategies for providing social housing or other social experience to the animals at the institution to ensure conformance with the Guide.
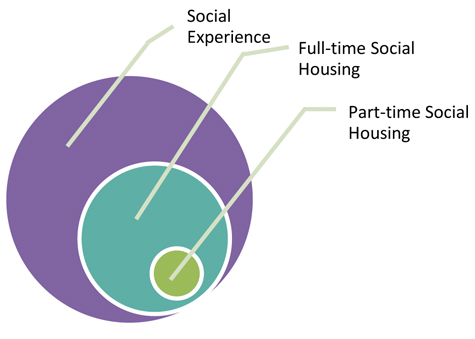
The relative social experience of a laboratory animal.
How often should vermin* traps (live trap or lethal) be checked by staff?
The Guide for the Care and Use of Laboratory Animals (Guide), NRC 2011 indicates that pest control managed by the animal care and use program personnel should be designed to prevent, control, or eliminate the presence of or infestation by pests in the animal environment. The Guide states (pg. 74) that "If traps are used, methods should be humane; traps that catch pests alive require frequent observation and humane euthanasia after capture" and also notes (pg. 112) that "all animals should be observed for signs of illness, injury or abnormal behavior by a person trained to recognize such signs. As a rule, such observation should occur at least daily." The Guide (pg. 105) further indicates that an adequate veterinary care program consists of assessment of animal well-being and effective management of pain and distress. The Guidelines of the American Society of Mammalogists for the Use of Wild Mammals in Research, an AAALAC International Reference Resource, indicate that lethal traps should result in a clean, effective kill and should be checked at least once a day, and in the event that an animal is still alive, it should be immediately dispatched in accordance with guidelines of the American Veterinary Medical Association.
In considering the information provided by the above references, the Council on Accreditation has determined that live mouse traps should be checked at least daily to prevent potential animal distress related to food or water deprivation. If the live trap provides food and water, then the IACUC should determine the appropriate frequency of checking traps. Alternatives to "sticky/adhesive" live board traps should be used for mice to avoid unnecessary animal distress as required by the Guide as part of a program of adequate veterinary care. Lethal traps (e.g., snap traps) should be checked daily so that in the event that a animal is still alive, it can be promptly euthanized. Council also considers that this guidance applies to all mice, be they laboratory or vermin mice.
*Excluding arthropods.
A researcher at our institution plans to use isopropyl alcohol to disinfect the surgical instruments she uses for a survival rodent surgical procedure. Will this be acceptable to AAALAC?
In 2001, AAALAC International published guidance on the use of alcohol as a skin disinfectant and for instrument sterilization (click here). At that time, AAALAC’s Council on Accreditation stated that the use of alcohol as a skin disinfectant for rodent survival surgery was acceptable, but that the blanket use of alcohol for surgical instrument preparation was not acceptable. The Guide for the Care and Use of Laboratory Animals (NRC 2011) upholds the position that, "Alcohol is neither a sterilant nor a high-level disinfectant." Recent evidence, however, does support that the use of alcohol may be acceptable for some procedures if prolonged contact times are used (Huerkamp 2002) or for limited numbers of serial rodent surgeries under specific conditions (Keen et al. 2010). The IACUC (or comparable oversight body) must evaluate the use of alcohol on a case-by-case basis with due consideration for animal welfare and scientific outcomes based on a review of current relevant literature, and consistent with expected surgical outcomes.
On page 128 of the Guide for the Care and Use of Laboratory Animals (NRC 2011), there is a new section which describes the importance of intraoperative monitoring during surgical procedures. The description includes evaluation of parameters such as anesthetic depth, body temperature, cardiac and respiratory rates and pattern as well as blood pressure. Will AAALAC now require that these parameters be monitored and recorded for all surgical procedures?
Site visitors will often evaluate protocols, standard operating procedures and surgical records to assess the appropriateness of anesthesia, analgesia, intraoperative surgical monitoring, post-operative care and outcome. As stated in the 1996 Guide and detailed more specifically in the 2011 Guide, the overall goal of surgical monitoring is to “increase the likelihood of a successful surgical outcome." With that goal in mind, the Council acknowledges that the level and detail required for intraoperative monitoring can vary with the species, invasiveness and complexity of the surgical procedure, duration of surgery and other factors, such as anesthetic regimen and the use of neuromuscular blockade. In general, the greater the potential for pain or distress, procedural complexity, duration, or likelihood of an unsuccessful outcome, the greater the need for detailed, intensive intraoperative monitoring. The Council expects that the level of intraoperative monitoring and recordkeeping will be based on these factors. For example, the level of intra-operative monitoring might be minimal for short, minor procedures on rodents. In contrast, extensive intraoperative monitoring would likely be required for long, complex, major procedures regardless of the species involved. Appropriate intraoperative monitoring for non-survival surgical procedures is also expected, using the above criteria. As always, the Council will evaluate the adequacy of intraoperative monitoring using a performance based approach which assesses whether procedures meet the goals and provide a successful outcome.
What level of monitoring and record-keeping are expected for rodent surgery?
There is clear general consensus in relevant resources (e.g. , the Guide for the Care and Use of Laboratory Animals (Guide), NRC 2011; Medical Records for Animals Used in Research, Teaching, and Testing: Public Statement from the American College of Laboratory Animal Medicine, ILAR 2007; Rodents: Laboratory Animal Management, NRC 1996; Research Animal Anesthesia, Analgesia and Surgery, SCAW 2007) that monitoring of rodents during surgery is critical so that animals are maintained under a surgical plane of anesthesia and that therapeutic intervention can be provided should unexpected physiological responses occur. Perioperative assessment of the physiological status (especially body temperature, but depending on other factors, also respiratory rate, heart rate, blood pressure, blood gases, ECG, etc.) and anesthetic depth are valuable metrics for this purpose. Monitoring is also key to ensuring that sound research data will ultimately be collected from the animals. The level of detail contained in the records should accurately reflect the monitoring being performed.
Therefore, while AAALAC does not have a policy that stipulates the level of documentation for surgical procedures, the Guide does recommend that pre-surgical planning include consideration of record-keeping, and AAALAC would expect that this would occur and that the level of monitoring and record-keeping would be adjusted to the type of procedure, health of the animal, etc. Good record-keeping is also important so the Institutional Animal Care and Use or Oversight Body (IACUC/OB) can track whether or not a specific animal had undergone more than one survival surgical procedure, as multiple survival surgical procedures need to be handled in a specific manner by the IACUC/OB. To summarize, then, there is no “cookie-cutter” approach to monitoring and documentation associated with surgical procedures, but AAALAC site visitors would expect all the factors described to be evaluated by the IACUC/OB for all surgical procedures when making these determinations.
The Guide for the Care and Use of Laboratory Animals (NRC 2011) NRC 2011 notes the ongoing controversy regarding the use of carbon dioxide (CO2) for euthanasia due to its aversive characteristics. What are AAALAC International's expectations on the use of CO2 for euthanasia?
The Guide states that "Unless a deviation is justified for scientific or medical reasons, methods should be consistent with the AVMA Guidelines on Euthanasia (AVMA 2007 or later editions)." The AVMA Guidelines for the Euthanasia of Animals: 2020 Edition, an AAALAC International Reference Resource, categorizes CO2 euthanasia as acceptable under certain conditions and states that "Carbon dioxide, with or without premedication with halogenated anesthetics, is acceptable for euthanasia of small rodents.” Also, “Carbon dioxide exposure using a gradual-fill method is less likely to cause pain due to nociceptor activation by carbonic acid prior to onset of unconsciousness; a displacement rate from 30% to 70% of the chamber volume/min is recommended for rodents….The practice of immersion, where conscious rodents are placed directly into a container prefilled with 100% CO2, is unacceptable." In addition, “Carbon dioxide and CO2 gas mixtures must be supplied in a precisely regulated and purified form without contaminants or adulterants, typically from a commercially supplied cylinder or tank....As gas displacement rate is critical to the humane application of CO2, an appropriate pressure-reducing regulator and flow meter or equivalent equipment with demonstrated capability for generating the recommended displacement rates for the size container being utilized is absolutely necessary."
However, as the Guide states (pp. 12-13), "The body of literature related to animal science and use of animals is constantly evolving, requiring Programs to remain current with the information and best practices." Therefore, the appropriate displacement rate for different rodent species may change as the science regarding CO2 euthanasia develops.
[1] This also applies to those rare cases when CO2 is used for anesthesia.
The 8th edition of the Guide for the Care and Use of Laboratory Animals (NRC 2011) has changed the guidance offered to institutions regarding the use of recycled air to ventilate animal rooms as compared to the recommendations in the 1996 Guide. Can we now relax our previously rigidly held standard that no more than 50% of the air delivered to an animal room can be recycled?
AAALAC International has determined that the discussion in the 2011 Guide regarding recycling air should be augmented with several points noted in the 1996 Guide. In general, the use of non-recycled air is preferred for ventilation of most animal use and holding areas. If recycled air is used, the exhaust air should be HEPA-filtered (high efficiency particulate air-filtered) to remove airborne particles before it is recycled; the extent and efficiency of filtration should be proportional to the estimated risk. Gaseous filtration, such as with activated charcoal filters, should also be considered when recycling air. The supply air should not exceed 50% recycled air and recycled air should be returned only to the room or area from which it was generated, except if it comes from other than animal housing areas and the source of the air poses no concerns for animal health. An exception to this recommendation would be mass air displacement clean-rooms, which generally use a minimum of 75% recirculated air due to their extremely high air exchange rates, which can be up to 700 air changes per hour for some clean-room classes. In all cases, the recycled air should be appropriately conditioned and mixed with sufficient fresh air to address air quality, thermal and humidity requirements of animals in the space. As noted in both the 1996 and 2011 editions of the Guide, the risk in some situations may be too great to consider recycling air.
The AAALAC International Program Description has a table (Appendix 11, Heating, Ventilation and Air Conditioning (HVAC) System Summary) where comprehensive HVAC data are requested. Can you clarify AAALAC’s expectations for HVAC performance data for buildings used in agricultural programs?
Proper air quality for animal facilities is important for the health and welfare of the animals as well as for the personnel working in the facilities. The Guide for the Care and Use of Agricultural Animals in Research and Teaching (Ag Guide), American Dairy Science Association®, the American Society of Animal Science, and the Poultry Science Association 2020, provides a description of Ventilation and Air Quality (p. 18) as well as guidance on methods and strategies to optimize the environment. In agricultural settings, the design and use of the buildings will determine the extent of HVAC information that is needed. In agricultural situations, the assessment of relative pressurization and air exchange rates may be unnecessary. However, pressurization is important for containment facilities at ABSL-3-Ag. The provision of emergency power to exhaust fans is critical for animal housing where mechanical ventilation is required to maintain air quality and environmental temperatures. AAALAC site visitors will consider the buildings’ design and function and utilize the recommendations of the Ag Guide when assessing the HVAC data and air quality for agricultural programs.
AAALAC International is a private, nonprofit organization that promotes the humane treatment of animals in science and education through a voluntary accreditation program and educational programs. More than 1,200+ animal care and use programs in 52 countries have been awarded AAALAC International accreditation, demonstrating their commitment to responsible animal care and use. These programs include academic institutions, commercial organizations, hospitals, and government agencies. AAALAC has been working to promote animal welfare and enhance research and education since 1965.
AAALAC International is governed by a Board of Directors, and advised by representatives from our Member Organizations. The Member Organizations represent more than 70 prestigious scientific, educational and professional organizations. Each of these Member Organizations appoints a Member Organization Delegate to serve a three-year term. By actively involving major organizations, AAALAC International remains responsive to the issues that members face, while making sure that members of the scientific community understand and support the AAALAC International accreditation program.
The Council on Accreditation is at the heart of AAALAC's mission and work. The Council is comprised of highly accomplished animal care and use professionals from around the globe who conduct program assessments that determine which institutions are awarded AAALAC accreditation. Their responsibilities include conducting site visits, reviewing site visit reports, evaluating information, reviewing yearly reports from accredited institutions, and conferring the accreditation status of institutions. The Council is divided into North American, European, and Asia-Pacific Sections.
Council members are typically selected from AAALAC's pool of Ad Hoc Visitor (see next question). Potential new members are nominated by current Council members, Board members, or colleagues from AAALAC Member Organizations. Potential new members are evaluated for their knowledge and experience with the site visit review process. Soft skills such as diplomatic communication and the ability to identify programmatic concerns are essential in a successful potential Council member. Council members are highly qualified and knowledgeable about diverse animal program and management issues. Council members serve 3 year terms which are renewable for a maximum of four terms.
AAALAC maintains a worldwide pool of more than 400 Ad Hoc Visitors who have expertise within the realm of traditional laboratory animal species as well as specific expertise (for example, in aquatics, wildlife, or agricultural science). Many also have unique discipline competencies, such as applied neuroscience, behavioral science, toxicology, pharmacology or physiology. Ad Hoc Visitors accompany Council members on site visits and make recommendations to the Council. These individuals add depth to the site visit team. They understand the intricacies of combining research, testing and educational missions with animal welfare.
Individuals who want to become an Ad Hoc Visitor submit an application. Once a year, a committee consisting of members of the Council reviews all applications and selects candidates based on established guidelines. Ad Hoc Visitors must have a minimum of four years of training or experience in the care or use of animals used in research and education. Their experience must show that they have acquired knowledge of performance standards and regulations. Ad Hoc Visitors serve three-year terms.
Currently, more than 1,200 animal care and use programs in 52 countries have earned AAALAC International accreditation. These programs include academic institutions, commercial organizations, agricultural research programs, government agencies, hospitals, nonprofit organizations, and biotechnology and pharmaceutical companies.
No, AAALAC is not a regulatory body and does not make or enforce regulations. Instead, AAALAC relies on widely accepted guidelines, such as the Guide for the Care and Use of Laboratory Animals (NRC 2011), and other resources. AAALAC does, however, publish "position statements" that can be used as supplemental guidelines in dealing with certain issues, such as the use of farm animals, occupational health and safety, or adequate veterinary care. AAALAC also publishes its "Rules of Accreditation," a document that lists the minimum criteria institutions must meet before they can apply for accreditation.
Does AAALAC International have a checklist, or other resources, that can be used when designing a new animal facility, or conducting a major renovation, to help ensure that the facility meets accreditation standards?
AAALAC International does not provide a design checklist, as we use a performance standard (outcome-based) approach to evaluate animal care and use programs during the accreditation process. This approach allows flexibility in facility design based on factors such as research scope, species housed, institutional goals, available resources, and geographic location. Because of this variability, a universal checklist isn’t practical. Instead, we recommend working with architects and consultants experienced in designing and operating animal facilities that align with your program’s specific needs and accreditation goals.
Learn more about our Performance Standard Approach.AAALAC evaluates all aspects of an animal care and use program. An animal program (as defined by AAALAC) includes an organization's procedures and overall performance in animal care and use. The basic components that are evaluated include (but are not limited to) institutional policies, animal husbandry, veterinary care, occupational health and safety, and the physical plant.
Yes. AAALAC encourages every animal program, large or small, to achieve the highest standards for responsible animal care and use. The standards used to evaluate programs are universal and can be implemented in programs of any size. Likewise, programs using nontraditional research animals, such as fish or birds, are also encouraged to seek accreditation.
Yes. Programs that use agricultural animals in research and education are embracing the AAALAC accreditation program. The Guide for the Care and Use of Agricultural Animals in Research and Teaching (Ag Guide), American Dairy Science Association®, the American Society of Animal Science, and the Poultry Science Association 2020, along with the Guide for the Care and Use of Laboratory Animals (NRC 2011), is used to provide general parameters for the use of agricultural animals. For animals in an agricultural setting, AAALAC International takes the position that, in accredited facilities, the housing and care for farm animals should meet the standards that prevail on a high-quality, well-managed farm. For further guidance please see our Position Statement on this subject.
Organizations seeking and maintaining accreditation are asked to pay a one-time application fee and subsequent annual fees. Both of these fees are based on a sliding scale that generally correlates with the size of an institution’s animal facility. An institution that has very few animals will pay much smaller fees than one that maintains a large vivarium and extensive support areas. The fees cover the cost of periodic site visits and administrative expenses.
AAALAC International determines the group classification during the review of the application. This classification, based primarily on the size of the facility and the time necessary to conduct a site visit, establishes a fair fee schedule. The established fee schedule may be amended as necessary to reflect the costs of operating the accreditation program. Revocation of accreditation shall be automatic if a unit is twelve (12) months in arrears for payment of fees. Please see http://www.aaalac.org/accreditation/fees-and-deadlines/ for the fee schedule.
Payment may be made by check, credit card, wire transfer (contact the AAALAC International office for bank details), or purchase order.
Organizations seeking accreditation are required to submit an application package which consists of a two-page application form, Instructions for completing and submitting the Program Description, and a template that will guide you through the process of preparing your “Program Description”. The Application form, Instructions and Program Description template can be found at https://www.aaalac.org/accreditation/apply-for-accreditation/. AAALAC conducts its evaluations on a trimester schedule. New applications undergo a pre-review process before being assigned a trimester to be visited. Organizations applying for accreditation are required to submit a non-refundable application fee following application approval and before the on-site assessment.
Deadlines for New Applications:
All organizations participating in the accreditation program pay an annual fee. After an organization earns accreditation, it must be evaluated every three years in order to maintain its accredited status. Currently more than 1,140+ organizations in 52 countries/regions have earned AAALAC accreditation.
What are the deadlines for submitting a Program Description to AAALAC International? When would the site visit occur?
| Trimester | Program Description Due Date |
Site Visit Occurs | Site Visit Report Preparation | Council Meeting |
| Summer | April 1 | May-July | August | September |
| Fall | August 1 | September - November | December | January |
| Winter | December 1 | January - March | April | May |
Organizations seeking accreditation are required to submit a non-refundable fee with the application materials. This fee covers the cost of the initial site visit. All applicants granted Provisional or Full Accreditation status pay an annual fee. Annual fees cover the cost of subsequent regularly scheduled triennial site revisits. The cost for interim site revisits conducted before the three-year interval, whether initiated by AAALAC International or the institution, will be borne by the institution.
AAALAC International determines the group classification during the review of the application. This classification, based primarily on the size of the facility and the time necessary to conduct a site visit, establishes a fair fee schedule. The established fee schedule may be amended as necessary to reflect the costs of operating the accreditation program. Revocation of accreditation shall be automatic if a unit is twelve (12) months in arrears for payment of fees. Please see http://www.aaalac.org/accreditation/fees-and-deadlines/ for the fee schedule.
Payment may be made by check, credit card, wire transfer (contact the AAALAC International office for bank details), or purchase order.
How does AAALAC ensure confidentiality?
Unlike many government regulatory systems, the entire accreditation process is confidential. The accreditation evaluation and its results are kept between the organization seeking accreditation and AAALAC International—even if deficiencies are found. AAALAC's purpose is to provide a peer-evaluation that results in valuable information organizations can use to improve their programs and achieve new levels of excellence. Board and Council members, Ad Hoc Visitors and AAALAC staff are all required to sign confidentiality agreements. Conflict-of-interest statements are also signed by each site visitor. AAALAC representatives agree to treat all materials as privileged, and safeguard the materials in their possession. Of course, accredited organizations are free to share their AAALAC reports if they choose to do so.
Our institution just received notice that AAALAC International will be conducting a “drop-in” visit. How does this differ from our routinely scheduled site visit and what are its implications?
Since 1989, and with endorsement of AAALAC’s Board of Directors, AAALAC has conducted drop-in visits—both “for cause” and “not-for-cause. ” While very rare, drop-in visits of either type may occur between the routinely scheduled triennial visits. In both cases, relatively short notice (one day to a few days) of the pending drop-in visit is provided. The scope of the drop-in visit is typically very focused; more rarely, the entire animal care and use program may be reviewed during a drop-in visit. Costs for the drop-in visit are absorbed by AAALAC.
Occasionally, a site visit team will conduct a drop-in visit to an institution in an area where a routinely scheduled visit is occurring at another organization. These visits may be prompted by significant programmatic changes that have occurred at the institution subsequent to the routine site visit, such as critical organizational changes, an about-face in a commitment made to Council (e.g. , re-opening an animal housing area that had been closed in response to a site visit observation), etc. Such “not-for-cause” visits provide the Council on Accreditation an update as to the status/functioning of the institution.
Drop-in visits “for cause” may be conducted following a meeting of the Council on Accreditation, during which the Council has determined that an issue identified during the regularly scheduled site visit was of sufficient seriousness that immediate follow-up by the Council, in the form of an additional on-site assessment, was necessary. Occasionally, a drop-in visit “for cause” may be initiated by the Executive Office following receipt of a verifiable allegation related to the animal care and use program at an accredited institution.
Reports of observations made during either type of drop-in visit are taken to the Council on Accreditation. The observations made during a “not-for-cause” drop-in often result in no action being taken by the Council regarding the institution’s accreditation status. Alternatively, the report may prompt the Council to require the institution to provide additional documentation regarding the matter, or the Council may determine that a full site visit needs to be scheduled earlier than the typical three-year interval. The observations made during a drop-in “for cause” visit may result in no change to the accreditation status of the institution or the institution may be placed on less than Full Accreditation pending correction of the issue.
The 2011 Guide, like its 1996 predecessor, distinguishes between and provides definitions for the terms "should" and "must" to provide the Guide Committee's interpretation of the relative importance of the recommendations made. It is not clear to me how these "shoulds" and "musts" translate into the mandatory items for correction and suggestions for improvement used by AAALAC International. Would you please clarify this?
The Guide states that, "Must indicates actions that the Committee for the Update of the Guide considers imperative and mandatory duty or requirement for providing humane animal care and use. Should indicates a strong recommendation for achieving a goal; however, the Committee recognizes that individual circumstances might justify an alternative strategy." Given the fundamental importance of the recommendations in the Guide that are prefaced with a "must," AAALAC's Council on Accreditation typically categorizes site visit findings that do not conform with a "must" statement in the Guide as a Mandatory item for correction. In AAALAC's nomenclature, a Mandatory item is a serious deviation from the recommendations of the Guide, and/or other AAALAC International standards, which has to be corrected to achieve or continue Full Accreditation. This judgment is based on the Council's assessment of the potential for the program deficiency to adversely affect the health, well-being or safety of animals or humans.
The second category of findings identified by AAALAC during the on-site assessments of animal care and use programs is comprised of Suggestions for Improvement (SFIs). These are recommendations that the Council on Accreditation feels are desirable to upgrade an already acceptable or even commendable program. SFIs are used to draw attention to recommendations that are typically denoted as "should" statements in the Guide. AAALAC considers the offering of SFIs to be an element of the peer review process that is designed to assist accredited programs by sharing the cumulative knowledge and experience of the Council. It should be noted that there is no obligation for institutions to make program changes based on suggestions for improvement; implementation of suggestions is, however, one means of promoting a high quality animal care and use program. Also, an SFI does not automatically become a Mandatory item for correction during the next site visit cycle if the same situation (e.g., procure, practice, etc.) is observed. However, if an issue is identified that is a "should" statement in the Guide, but is one of numerous issues noted within the same program area that collectively signal a broader problem, then it may be wrapped into a Mandatory item for correction.
It is also worthwhile to note that several requirements in the Guide are not prefaced with the word "must." Other terminology is occasionally used to convey the same level of imperative for complying with the statement. Examples include:
How does the Council on Accreditation ensure consistency in the application of standards and expectations for animal care and use programs? Can you provide information on how consistency is maintained for AAALAC accreditation of diverse programs around the world?
Consistency in program review is paramount to the AAALAC International accreditation process. As such, the Council on Accreditation has implemented procedures to promote consistency and identify and correct inconsistencies. Findings that are presented at site visit exit briefings are considered preliminary with final decisions made during the Council deliberations. If a finding is subsequently determined by the Council to be inconsistent with the Three Primary Standards, Reference Resources or applicable legislation*, a retraction or clarification statement is typically included in the accreditation status letter. This statement describes the Council’s final decision and how the finding was modified. Additionally, a standing committee of the Council reviews all findings that were judged to be inconsistent with AAALAC’s standards and provides guidance to the site visitors for future reference and continuing education.
*Because animal care and use oversight legislation varies around the globe, AAALAC International acknowledges performance-based approaches (Guide pp. 6-7) that allow flexibility while ensuring outcomes that meet the principles described in the Three Primary Standards.
There are three types of reporting requirements: Administrative Reports, Annual Reports, and Adverse Event Reports. Each is outlined below:
Administrative Reports
Administrative Reports should be submitted to AAALAC via email to accredit@aaalac.org.
What types of administrative reports must be reported promptly to the AAALAC International Executive Office?
Annual Reports
The Unit Contact of record’s email address is the only email address that is recognized by the AAALAC website to gain access to complete and submit the Annual Report.
Are there specific due dates for the Annual Reports?
Each year in mid-December, the AAALAC International office makes available the online Annual Report form. There are no specific due dates for submitting an Annual Report. However, AAALAC International’s Rules of Accreditation require that you submit an Annual Report in order to maintain your accreditation. An institution may choose from a variety of reporting periods (e.g., University fiscal year, calendar year, federal government fiscal year, government oversight body reporting period, etc.) as the AAALAC International reporting period. Your Annual Report must then be submitted within three months of the end of your chosen reporting period. Please be sure that the period covered is continuous with previous reports (i.e., there are no gaps and all periods are covered by a report).
What type of information is provided in the Annual Report?
Annual Reports should provide notification of any:
Yes, but remember we also need animal numbers for species not regulated by the USDA.
Adverse Event Reports
I am unclear what types of adverse events need to be reported. I would appreciate additional information to assist my institution in meeting this reporting requirement.
AAALAC recognizes that there have been many questions regarding when a unit is required to report a significant adverse event to the AAALAC office and that this has resulted in variability in reporting practices. The revised Guidance Statement clarifies expectations about reporting to the AAALAC office and emphasizes institutional responsibility for identifying, reviewing, and managing adverse events through strong local oversight and investigation. These institutional processes will continue to be evaluated during AAALAC site visits.
With the new Guidance Statement, AAALAC expects institutions to only notify the AAALAC Office when an event presents potential external, regulatory, or legal risk. This administrative change is intended to ensure constructive communication during situations that may affect institutional or AAALAC integrity while reinforcing accountability at the institutional level.
Please refer to the revised Guidance Statement “Adverse Events” for complete details on assessing and reporting adverse events. The Adverse Events Reporting FAQs provide additional information regarding this important change.
I cannot access the "Unit Login" area. How do I get the username and password?
The username and password is provided only to the designated Unit Contact at your institution. This individual may share this information with other members of the institution.
Our institution is preparing for its next AAALAC International site visit. Does AAALAC provide any information or guidance regarding common suggestions or findings that might help us with our preparation?
In addition to the educational materials posted on the AAALAC International website, AAALAC periodically reviews and analyzes information collected during accreditation site visits. These reviews identify program areas that receive the most "Suggestions for Improvement" or require "Mandatory" changes before accreditation can be granted. The most recent review looks at findings collected during site visits over a five-year period (2013-2018) and highlights challenge areas. Institutions can use this “Trends Data Review” as an educational resource or to assist with program evaluation and assessment, especially in preparation for an accreditation site visit..
1. Are institutions still required to investigate adverse events and noncompliance incidents?
Yes. Institutions are expected to continue identifying, investigating, and reviewing all adverse events and noncompliance incidents within their programs, especially those impacting animal welfare or human health. Adverse events and noncompliance incidents should be reviewed by the Institutional Animal Care and Use Committee (IACUC) or a comparable Oversight Body (OB), evaluated to determine root cause, addressed through appropriate mitigation strategies, and monitored as necessary. The outcomes of these processes should be thoroughly documented. These expectations have not changed.
2. What types of events should be reported to the AAALAC Office?
Reporting adverse events have always been part of the Rules of Accreditation, and as such, institutions are expected to promptly notify the AAALAC Office of events that may present external, regulatory, or legal implications. Examples include investigations by external oversight bodies or regulatory agencies, public records requests involving AAALAC documents, lawsuits alleging concerns related to the animal care and use program, events anticipated to attract media or activist attention, or other situations that may create risk to the integrity of the institution or AAALAC. If there is uncertainty about whether a situation should be reported, institutions are encouraged to contact the AAALAC Office for guidance.
3. If institutions are no longer expected to report every adverse event to the AAALAC Office, do institutions still need an adverse event assessment and reporting plan?
Yes. Each accredited unit should develop and maintain a process or plan that defines adverse events and noncompliance incidents and describes how they are assessed and managed. This plan should include procedures for risk assessment, mitigation, and reporting to appropriate internal and external oversight bodies (including AAALAC International), as applicable. The effectiveness of this process will continue to be evaluated by AAALAC Council during site visits.
4. If institutions are no longer expected to report every adverse event to the AAALAC Office, must events still be documented?
Yes. Institutions are expected to maintain appropriate documentation of adverse events, such as meeting minutes, reports, or tracking systems. During a site visit, the Site Visit Team may review documentation to understand what occurred, how the event was investigated, what corrective or preventive actions were implemented, how the risk of recurrence was addressed, and whether trends are monitored over time. Maintaining clear and thorough documentation remains essential.
5. Should unexpected animal deaths or procedural complications be reported to AAALAC?
Adverse events and noncompliance incidents impacting animal welfare or human health that are appropriately identified, investigated, and mitigated through institutional processes do not need to be reported to the AAALAC Office. However, reporting is expected if such events lead to external investigations, legal action, or circumstances that may result in media attention or external risk.
6. How will AAALAC Site Visit Teams evaluate adverse events?
Site visit teams will continue to assess how institutions identify and manage adverse events within their programs. This includes evaluating the role of the IACUC/OB in reviewing events, the use of root cause analysis, the implementation of mitigation strategies, and the monitoring of trends over time. The site visit teams will evaluate documentation supporting the institution’s adverse event processes during the site visit. The overall emphasis remains on effective oversight and a demonstrated Culture of Care.
7. Does this change reduce expectations for compliance?
No. This clarification does not represent a reduction in expectations or standards. Institutions remain responsible for protecting animal welfare, safeguarding human health, maintaining integrity, and demonstrating effective oversight. The revision pertains only to which events should be reported to the AAALAC Office.
8. What should institutions do if they are unsure whether an event should be reported?
If there is uncertainty regarding whether an event should be reported or about the timing of reporting, institutions should contact the AAALAC Office for guidance. AAALAC encourages institutions to seek clarification when questions arise. Options include contacting the main office at +1-301-696-9626 or accredit@aaalac.org, contacting regional office staff, or using the online portal in the Unit Login area of the website to request a phone call.
9. AAALAC requires institutions to report public records requests involving AAALAC documents. Our institution receives frequent FOIA or public records requests. Should all of these be reported?
The key consideration is the potential for exposure beyond the institution or impact, rather than the number of requests received. Institutions that regularly receive FOIA or other public records requests and manage them through established procedures without escalation or increased risk do not need to report those requests routinely to AAALAC. The AAALAC Office should be notified when these document requests include requests for copies of the program description or any correspondence between the unit and AAALAC International.
10. What is the key takeaway for institutions?
Institutions are expected to maintain strong local/institutional oversight of adverse events by identifying and investigating events, documenting findings, implementing appropriate mitigation strategies, and escalating concerns to the AAALAC Office when external, regulatory, or legal risk may be present.
11. In the past, I have been advised that if an adverse event is reported to NIH/OLAW or USDA (both of which are subject to FOIA), you should let AAALAC know so that they are aware in advance.
It is not necessary to copy AAALAC on adverse event reports that are submitted to NIH/OLAW or the USDA in accordance with the adverse reporting expectations for these regulatory agencies.
12. If we report an adverse event to NIH/OLAW or the USDA, is this considered an "investigation by an external agency?"
No. When you report an adverse event to a regulatory agency (e.g. NIH/OLAW or USDA in the United States of America), this is considered self-reporting and is not considered an investigation by an external oversight body.
13. We just had our annual USDA inspection and received a citation. Is this considered an investigation by an external oversight body?
No. The results of the USDA inspection are not considered an investigation by an external oversight body. However, if the USDA opens an investigation following an annual inspection, this would need to be reported to AAALAC.
14. To what risk is our accreditation if we do not successfully predict what may or may not go public and create media attention or trigger an investigation by an external body?
AAALAC understands that it can be difficult to determine if an adverse event will result in media attention or trigger an investigation by an external body. As soon as you become aware that the adverse event is now garnering this level of attention, please inform AAALAC.